FAQ
Product information, usage, and contact-related common explanations
- QCT
- Osteoporosis
- Traditional Method for Bone Density Assessment - DXA
- Bone’s QCT
- Comparative Analysis of QCT Products
- Clinical Application Data Transmission
- Pricing and Medical Insurance
- Bone’s Manufacturer
- Other FAQ
-
QCT
-
What is QCT?
QCT is a new-generation device for measuring bone mineral density. It performs precise measurements through 3D images generated by CT scans. The measurement results can be used for diagnosing osteoporosis. The commonly measured clinical regions include the hip and lumbar spine.
-
Which patients are recommended to undergo bone density measurement using QCT?
(1) Patients diagnosed with osteoporosis or at risk of low bone mass.
(2) Patients currently undergoing treatment for osteoporosis and requiring regular monitoring.
(3) Patients receiving or planning to receive steroid therapy.
(4) Patients with advanced hyperparathyroidism.
(5) Patients with abnormal vertebral morphology.
(6) Patients with exceptionally small or tall body frames.
(7) Patients with obesity.
(8) Patients presenting with degenerative spinal diseases.
-
What are the clinical contraindications for the examination?
(1)Patients who have recently undergone examinations involving contrast agents, iodine, or other reagents.
(2)Women who are pregnant or possibly pregnant.
-
Which clinical guidelines are followed for this examination?
(1)Chinese Quantitative CT Osteoporosis Diagnosis Guidelines (2018), published in the Chinese Journal of Osteoporosis and the Chinese Journal of Health Management
(2)Chinese Guidelines for the Diagnosis and Treatment of Osteoporosis in the Elderly (2018)
(3)Primary Osteoporosis Diagnosis and Treatment Guidelines (2022)
(4)Expert Consensus on the Imaging and Bone Density Diagnosis of Osteoporosis (2020)
(5)The International Society for Clinical Densitometry (ISCD) Expert Consensus (2019)
(6)American College of Radiology (ACR) Clinical Guidelines (2018)
(7)ISCD Expert Consensus (2019)
(8)American College of Radiology Clinical Guidelines (2018)
-
Which departments can issue QCT orders?
Orthopedics, Endocrinology, Rheumatology, Geriatrics, and Physical Examination Departments
-
Is there a standard for QCT diagnosis?
The American College of Radiology has recognized QCT as a method for measuring bone density and has established diagnostic criteria.
Diagnosis is based on the average QCT bone density of the cancellous bone in the lumbar vertebrae (typically the 1st and 2nd lumbar vertebrae): QCT bone density >120 mg/cm³: Normal bone density; QCT bone density between 80–120 mg/cm³: Low bone mass; QCT bone density <80 mg/cm³: Osteoporosis. For the hip bone, QCT bone density follows the World Health Organization (WHO) T-score standards; osteoporosis is diagnosed when the lowest T-score of the total hip or femoral neck corresponds to a value of ≤-2.5
-
Osteoporosis
-
What is osteoporosis?
(1)Osteoporosis is the most common bone disease and is characterized as a systemic skeletal condition marked by low bone mass and microarchitectural deterioration of bone tissue. This leads to increased bone fragility and a higher risk of fractures.
(2)In 2001, the National Institutes of Health (NIH) defined osteoporosis as a skeletal disorder characterized by reduced bone strength and increased fracture risk. While low bone mass is a major risk factor for osteoporotic fractures, other risk factors also contribute.
(3)Osteoporosis can occur at any age but is most commonly seen in postmenopausal women and elderly men.
(4)Osteoporosis is classified into primary and secondary types.
Primary osteoporosis includes:
i.Postmenopausal osteoporosis (Type I): Typically occurs within 5–10 years after menopause in women.
ii.Senile osteoporosis (Type II): Generally refers to osteoporosis occurring after the age of 70.
iii.Idiopathic osteoporosis: Predominantly affects adolescents, with an unclear cause.
Secondary osteoporosis: Refers to osteoporosis caused by any disease, medication, or other identifiable condition that affects bone metabolism.
-
What is the current status of osteoporosis in China?
With the intensifying aging population, osteoporosis has become a significant public health issue in China, showing a sharply increasing prevalence trend.
(1)Large number of affected individuals: The prevalence of osteoporosis among people aged 50 and above in China is 19.2%, rising to 32.0% among those aged 65 and above. It is estimated that there are approximately 90 million osteoporosis patients nationwide, with women comprising the vast majority (about 70 million). Among women aged 65 and above, the prevalence exceeds half the population (51.6%).
(2)Severe consequences: Osteoporotic fractures are the most serious complications of osteoporosis and are one of the leading causes of disability and mortality in elderly individuals.
(3)Heavy economic burden: It is projected that by 2035, China’s medical expenses for major osteoporotic fractures (wrist, vertebral, and hip fractures) will reach 132 billion RMB and will further climb to 163 billion RMB by 2050.
Reference:
Chinese Guidelines for the Diagnosis and Treatment of Osteoporosis in the Elderly (2022)
-
What are the risk factors for osteoporosis and fragility fractures?
Among populations with the following conditions:
(1)Low body weight (BMI < 20 kg/m²)
(2)Excessive alcohol consumption (> 2 units/day)
(3)High sodium intake
(4)Low bone density
(5)Inadequate calcium and/or vitamin D intake
(6)Prolonged immobilization
(7)Smoking
(8)Decreased physical activity
(9)History of falls
Among populations with the following diseases:
(1)Endocrine disorders: Diabetes, hyperparathyroidism, hyperthyroidism, primary hyperparathyroidism, hypopituitarism, hypogonadism, Cushing’s syndrome, anorexia nervosa, androgen insensitivity syndrome, hypercalciuria, etc.
(2)Rheumatological and autoimmune diseases: Rheumatoid arthritis, systemic lupus erythematosus, ankylosing spondylitis, and other rheumatic diseases.
(3)Gastrointestinal diseases: Inflammatory bowel disease, malabsorption, chronic liver disease, gastrointestinal bypass or other surgeries, pancreatic disease, and celiac disease.
(4)Neuromuscular diseases: Epilepsy, Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, stroke, spinal cord injury, and muscular dystrophy.
(5)Hematological diseases: Multiple myeloma, lymphoma, leukemia, monoclonal gammopathy of undetermined significance (MGUS), hemophilia, sickle cell anemia, systemic mastocytosis, thalassemia, etc.
(6)Other conditions: Moderate to severe chronic kidney disease, asthma, chronic metabolic acidosis, chronic obstructive pulmonary disease (COPD), post-organ transplantation, congestive heart failure, depression, HIV/AIDS, amyloidosis, etc.
Among populations taking the following medications:
Gonadotropin-releasing hormone (GnRH) agonists, glucocorticoids, anticoagulants (e.g., heparin), proton pump inhibitors, long-term antidepressants, antiepileptics, thiazolidinediones (TZDs, insulin sensitizers), aromatase inhibitors, cancer chemotherapy drugs, barbiturates, aluminum-containing antacids, cyclosporine A, tacrolimus, thyroid hormones, selective serotonin reuptake inhibitors (SSRIs), and antiviral drugs.
Recommendations from the 2017 Primary Osteoporosis Diagnosis and Treatment Guidelines:
In medical institutions equipped with DXA or QCT, it is recommended to combine bone fracture history with annual DXA or QCT tests. Bone turnover biochemical markers should be assessed every 3–6 months to monitor anti-osteoporosis treatment efficacy (Grade 2B recommendation).
Screening recommendation (Grade B): Women aged 65 and above, and postmenopausal women under 65 who are at high risk for osteoporosis.
-
What are the typical symptoms of osteoporosis?
(1)Pain: Patients may have lower back pain, generalized bone pain, or skeletal pain, which worsens with increased load or restricted movement. In severe cases, turning over, sitting up, or walking can become difficult.
(2)Spinal deformity: Severe osteoporosis can cause height reduction, kyphosis, spinal deformities, and limited extension. Compression fractures of the thoracic vertebrae may lead to chest deformities, affecting heart and lung function, while lumbar fractures can alter abdominal anatomy, resulting in symptoms such as constipation, abdominal pain, bloating, reduced appetite, and premature satiety.
(3)Fragility fractures: Fragility fractures occur under low-energy or non-violent events, such as minor falls or routine daily activities. Common fracture sites include the thoracic spine, lumbar spine, hip, distal radius/ulna, and proximal humerus. After experiencing one fragility fracture, the risk of subsequent fractures is significantly increased.
-
How does osteoporosis affect surgery?
Osteoporosis can significantly impact the outcomes of surgeries involving the spine, joints, and fractures. Clinical data show that, in patients with osteoporosis, the loosening rate of internal fixation after spinal surgery can reach up to 60%, and the graft nonunion rate can be as high as 50%. Additionally, osteoporosis increases the risk of complications such as loosening of internal fixation, pseudarthrosis, cage subsidence, fractures of adjacent vertebrae, and proximal junctional kyphosis (PJK) following spinal surgery.
-
Traditional method for bone mineral density testing -- DXA
-
What are the common methods for bone density measurement?
The common methods for measuring bone density include dual-energy X-ray absorptiometry (DXA), ultrasound bone densitometry, and quantitative computed tomography (QCT). Among these, ultrasound bone densitometry is used only for screening due to its limited accuracy, and DXA also has certain limitations. Currently, clinical guidelines recommend QCT and DXA as standardized and reliable methods for measuring bone density. For more details about the limitations of DXA, refer to section 3.2.
-
What are the issues with the DXA bone density measurement method? Is there clinical evidence?
(1)DXA cannot distinguish between cortical and cancellous bone: Cancellous bone is lost earlier and more quickly
(2)DXA cannot differentiate normal bone from degenerative structures: Patients with spinal degeneration may have osteophyte formation
(3)In elderly individuals, DXA measurements are on average 15% higher than the true value, with a diagnostic accuracy of only 60%
(4)The degree of degeneration from L1 to L4 progressively increases, with T-scores for L1 being relatively accurate
(5)In the presence of severe osteophytes, the likelihood of a positive result on QCT but a negative result on DXA is 3.649 times higher compared to cases without such factors

In the presence of severe osteophytes, the likelihood of a positive result on QCT but a negative result on DXA is 3.649 times higher compared to cases without such factors
(1) Lin W, He C, Xie F, et al (2023) Discordance in lumbar bone mineral density measurements by quantitative computed tomography and dual-energy X-ray absorptiometry in postmenopausal women: a prospective comparative study. Spine J 23:295–304. https://doi.org/10.1016/j.spinee.2022.10.014
(2)Kim M-W, Lee D-H, Huh J-W, Bai J-W (2022) The impact of obesity on the accuracy of DXA BMD for DXA-equivalent BMD estimation. BMC Musculoskeletal Disorders 23:1130. https://doi.org/10.1186/s12891-022-06076-0
(3)Choksi P, Jepsen KJ, Clines GA (2018) The challenges of diagnosing osteoporosis and the limitations of currently available tools. Clin Diabetes Endocrinol 4:12. https://doi.org/10.1186/s40842-018-0062-7
(4)Yu EW, Bouxsein ML, Roy AE, et al (2014) Bone loss after bariatric surgery: discordant results between DXA and QCT bone density. J Bone Miner Res 29:542–550. https://doi.org/10.1002/jbmr.2063
(5)Li N, Li X, Xu L, et al (2013) Comparison of QCT and DXA: Osteoporosis Detection Rates in Postmenopausal Women. International Journal of Endocrinology 2013:1–5. https://doi.org/10.1155/2013/895474
(6)Nazarian A, Cory E, Muller R, Snyder BD (2009) Shortcomings of DXA to assess changes in bone tissue density and microstructure induced by metabolic bone diseases in rat models. Osteoporos Int 20:123–132. https://doi.org/10.1007/s00198-008-0632-0
-
Why use QCT if DXA is considered the gold standard?
DXA is regarded as the traditional gold standard primarily because it was introduced earlier and was quickly incorporated into clinical guidelines. However, it has certain limitations in terms of clinical accuracy and diagnostic efficiency. In contrast, QCT has independent diagnostic criteria and the ability to directly diagnose osteoporosis, making it a more precise and reliable method in many cases.

Reference: Chinese Guidelines for the Diagnosis of Osteoporosis by Quantitative CT (2018)
Experts now regard QCT as the new generation gold standard for bone density testing, and there are even research studies and literature reports advocating for QCT to be recognized as the gold standard.
Kulkarni AG, Thonangi Y, Pathan S, et al (2022) Should Q-CT Be the Gold Standard for Detecting Spinal Osteoporosis? Spine (Phila Pa 1976) 47:E258–E264. https://doi.org/10.1097/BRS.0000000000004224
-
What are the main advantages of QCT compared to traditional DXA?
(1)Greater Precision: DXA measures 2D areal bone density, which is easily affected by bone degeneration, calcification, and other interferences, leading to significant errors; QCT measures 3D volumetric bone density, enabling differentiation between normal bone and degenerative bone, with reproducibility as high as 99%, making diagnosis and treatment monitoring more accurate.
(2)Broader Applications: QCT is suitable for patients with conditions such as osteoarthritis, scoliosis, degeneration, aortic calcification, and obesity, which DXA cannot reliably assess.
(3)Higher Efficiency: QCT is simple to operate, capable of delivering reports within 15 seconds, without requiring dedicated equipment or personnel, significantly reducing patient wait times.
(4)More Cost-effective and Practical: QCT can utilize previous CT data without additional radiation exposure, and one scan can provide both a lung screening and a bone density report simultaneously.
(5)More Advanced Technology: DXA is based on 1980s technology with limited application potential, whereas QCT represents a next-generation technology with higher clinical and research value.
-
Bone’s QCT
-
What modules does Bone’s QCT include?
Spinal bone density measurement, hip bone density measurement, localized bone density measurement, bone void analysis, soft tissue fat content measurement, CT liver-to-spleen ratio analysis, and body fat analysis.
-
How is Bone’s QCT applied clinically?
Bone’s QCT has been widely adopted by over 500 hospitals across China, including Peking Union Medical College Hospital, Peking University Third Hospital, and the General Hospital of the People’s Liberation Army. For example, at the Shanghai Workers' Sanatorium, it is used more than 1,000 times per month, enabling fast and accurate bone density assessments to support clinical diagnosis and health management.
-
Has Bone’s QCT been fully validated?
The Bone’s QCT product has undergone multicenter clinical trials and quality inspections by the National Medical Products Administration (NMPA) and is NMPA-certified. Test results show that the product has a reproducibility of up to 99%, with an absolute error of less than 1 mg/cm³.
The product's core algorithm and theoretical foundation have been recognized with technical patents and a National Science and Technology Progress Award (second prize).
During its development, Bone’s phantommeless QCT relied on tens of thousands of research data points. Since its market launch in 2022, the product has established scientific research collaborations with several tertiary hospitals and successfully published multiple academic papers within two years. The product has been fully validated and meets clinical expectations.

-
Can Bone’s QCT measurement results serve as a basis for clinical diagnosis?
Yes, Bone’s QCT measurements can serve as a basis for clinical diagnosis. The product has undergone prospective, multicenter clinical trials comparing its results with the international gold standard Mindways, demonstrating consistent and clinically equivalent outcomes. It provides osteoporosis diagnoses directly in accordance with the "Expert Consensus on Imaging and Bone Density Diagnosis of Osteoporosis (2020)" lumbar QCT diagnostic criteria for osteoporosis.
Additionally, Bone’s QCT holds a medical device registration certificate issued by the National Medical Products Administration (NMPA), complying with the requirements of Announcement No. 104 (2017) for the "Medical Device Classification Catalogue." It is classified as a Class II 21-04 medical software for decision-support purposes. Its officially intended use allows analysis of imaging or data to provide clinical reference values. Final diagnostic conclusions should be made by physicians, who integrate the product's clinical reference values with the patient’s fracture history and other clinical information.
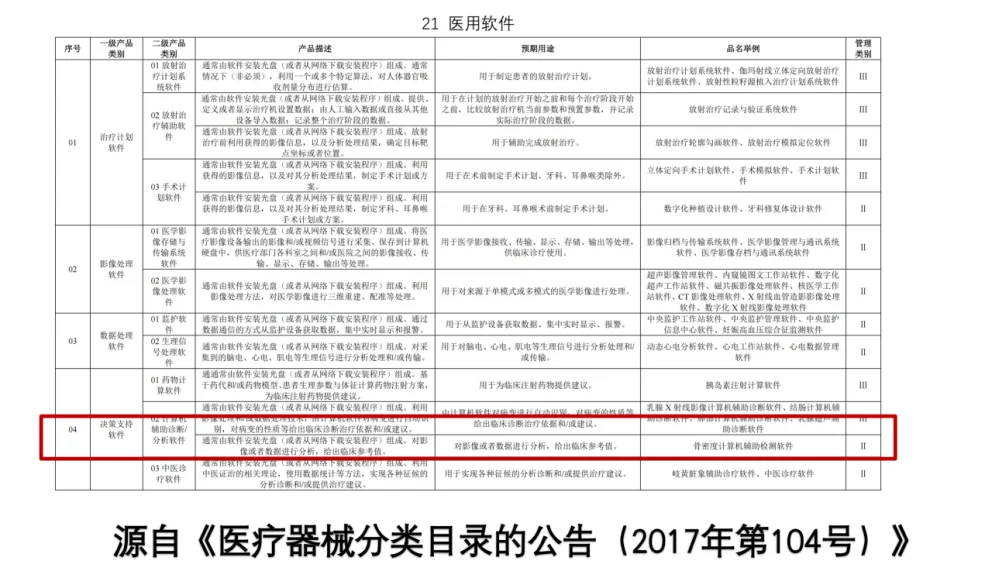
-
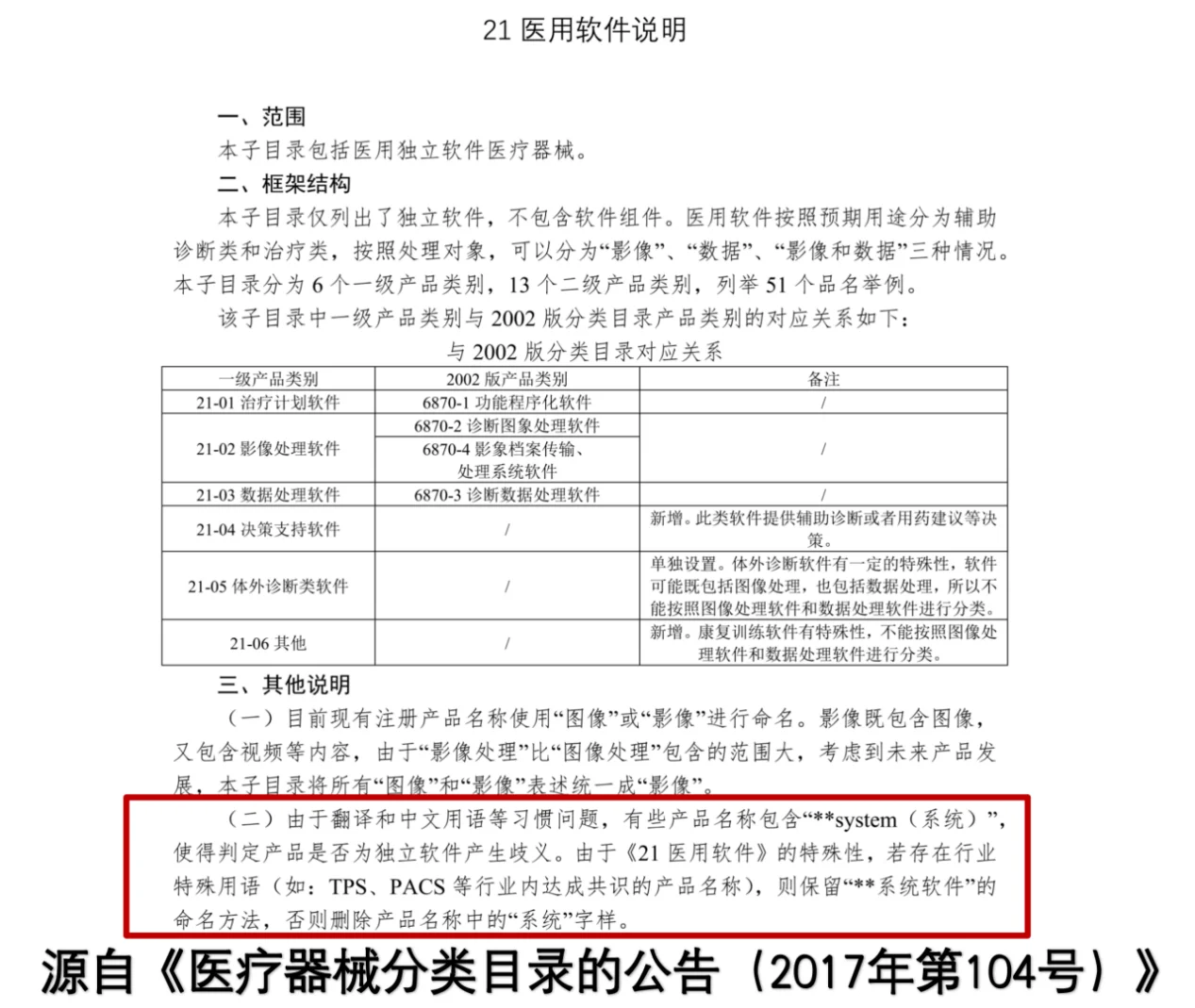
Is a QCT product named "System" superior to one called "Software"?
Products named as “system” and “software” both belong to Class II medical devices under the 21-04 category of medical software for decision support. Essentially, they are identical in function, as both can analyze images or data to provide clinical reference values. The designation “system” does not imply superiority.
The official document Announcement on the Medical Device Classification Catalogue (2017 No. 104) clarifies: “Due to differences in translation and conventions in Chinese terms, some product names include the word ‘system,’ creating ambiguity about whether the product is standalone software. For the special category of '21 Medical Software,' if there are industry-recognized terms (e.g., TPS, PACS), the naming convention of 'system software' is retained. Otherwise, the term 'system' is removed from the product name.”
-
Does the self-referencing phantom calibration technique used by Bone's QCT introduce errors because muscle and fat densities vary among individuals?
Currently, QCT technology on the market employs two calibration methods. The traditional approach relies on external phantom calibration, which uses the density reference values from a physical phantom to calibrate and determine the bone density reference values.
Bone's QCT uses a self-referencing phantom calibration technique, similar to external phantom calibration products, by utilizing areas with constant density reference values to calculate bone density reference values for the target bone region.
The difference lies in the reference material used. Instead of external phantoms, Bone's QCT uses the body's own muscle and fat regions as density references. A common misconception is that non-homogeneous regions, such as fat-infiltrated muscles, are used for calibration. This is incorrect. Bone's QCT exclusively relies on pure and stable muscle and fat regions with constant density reference values. The core algorithm and calibration methodology have been reviewed and approved by the National Medical Products Administration (NMPA), ensuring its reliability and safety for clinical application. It can be used with confidence in medical practice.
-
How is body fat segmented?
We collaborated with a team of experienced clinicians who manually annotated CT data layer by layer. These annotations were then used to train an artificial intelligence algorithm capable of segmenting subcutaneous, visceral, and muscle regions. After the algorithm completes the segmentation of target regions, optimization algorithms are used to determine fat threshold ranges, allowing for the distribution of fat in subcutaneous, visceral, and muscle regions to be determined. This results in a comprehensive fat volume analysis.
The body fat measurement functionality of Bone's QCT demonstrates high reproducibility, with an error rate of less than 1%. While the industry average segmentation accuracy is 0.8, Bone's QCT achieves a segmentation accuracy of 0.9, exceeding industry standards.
-
Comparative Analysis of QCT Products
-
What sets Bone's QCT apart?
(1)Precision: The only QCT device in China validated through multicenter clinical trials, equipped with a Chinese-specific database, a verified quality control system, and compatibility with all CT machine models for reliable results
(2)Efficiency: Skilled technicians generate reports in 15 seconds, with reusable test results allowing multiple analyses from one test
(3)Convenience: Data can be loaded and reports saved easily, with the capability to transmit reports back to PACS or print them conveniently
(4)Comprehensiveness: Provides full coverage of spine and hip measurements, offers rich research modules, and supports low-dose CT for measurements of bone density, sarcopenia, fatty liver, and body fat content, with the ability to analyze historical data
(5)Traceability: Ensures each test is traceable through synchronized calibration
(6)Flexibility: Supports various scanning parameters (current, voltage) and multiple CT machines, with flexible deployment options, including single and multi-station versions, meeting clinical needs and allowing for product customization
(7)Profitability: Achieves fast cost recovery with low hardware maintenance costs
(8)Innovation: Integrates over 30 years of orthopedic clinical, research, and industry experience, combining artificial intelligence and biomechanics to achieve precise local bone density measurement using synchronized self-referencing phantom calibration
-
Is Bone's QCT inaccurate without a phantom? How can it be used without one?
(1)Bone's QCT does have a phantom, utilizing an innovative and patented synchronized self-referencing phantom calibration technology
(2)This technology has been approved through quality inspections by the National Medical Products Administration (NMPA)
(3)It has also been validated through multicenter clinical trials, proving its scientific rigor and clinical reliability
(4)The scientific basis of Bone's QCT has been published in a core orthopedic journal, demonstrating the technical optimization and clinical applications: Liu Z-J, Zhang C, Ma C, et al (2022) Automatic phantom-less QCT system with high precision of BMD measurement for osteoporosis screening: Technique optimisation and clinical validation. Journal of Orthopaedic Translation 33:24–30. https://doi.org/10.1016/j.jot.2021.11.008
-
What are the advantages of Bone's QCT compared to competing products with external phantoms?
Competing products with external phantoms have the following limitations:
(1)Device Binding Restrictions: Products with phantoms are typically bound to a single CT device, lacking the flexibility of Bone's QCT, which can connect to multiple devices.
(2)Cumbersome Operations: External phantoms require regular/synchronized calibration, making maintenance complicated.
(3)Poor Scalability: Even asynchronous phantoms (asynchronous calibration) are usually limited to calibration for a single device, restricting their use in broader clinical scenarios.
In contrast, Bone's QCT adopts synchronized self-referencing phantom calibration technology, eliminating the need for an external phantom. It is not limited by the number of CT devices and offers greater flexibility and convenience.
-
QCT's radiation dose is too high; can Bone's QCT even be compared to DXA?
Bone's innovative self-referencing phantom QCT can reuse patients' existing CT data without adding extra radiation exposure. Combined with modern low-dose CT scans used for lung screening, Bone's QCT enables simultaneous and convenient bone density assessments with no additional radiation.
-
If the price is similar, why not choose American products?
According to relevant notices from the General Office of the State Council, domestic product standards must be implemented in government procurement, and products must be manufactured within China. Additionally, imported equipment often suffers from a lack of after-sales support and faces risks from geopolitical tensions and trade barriers, making the supply and maintenance services unreliable and unstable.

-
Are QCT's other functions recognized by clinical experts?
Globally, Professor Harry K. Genant is a highly influential pioneer in the field of QCT technology. His groundbreaking research on dual-energy CT quantitative bone mineral analysis, published in 1977, laid a solid academic foundation for QCT technology.
In China, QCT research and application have developed early, with steady progress in technological adoption and clinical practice. The large-scale use of QCT in orthopedics, along with its industry influence, has been driven by leading domestic orthopedic experts such as Tang Peifu, Wang Yipeng, Qiu Guixing, Ma Xinlong, Yang Huilin, Zhao Yu, and others.
We have established in-depth academic and clinical collaborations with these top Chinese experts. Under their leadership and promotion, QCT technology has been comprehensively standardized, efficiently implemented across clinical settings nationwide, and has led in the development of important clinical guidelines and expert consensus, such as the Chinese Guidelines for the Diagnosis and Treatment of Elderly Osteoporosis and the Clinical Guidelines for Bone Quality Assessment and Management in Orthopedic Surgery Patients. This has firmly established QCT's authoritative position and core value in orthopedic clinical applications.
-
Is Bone's QCT the same as the so-called “phantom-less” pure AI-based bone density products on the market? What are the differences?
Not at all. Pure AI-based products are fundamentally different from ours. They are often inaccurate and do not conform to clinical diagnostic standards.
(1)Different Principles: AI-based bone density products are less accurate
i.Pure AI Products (Guessing Data): These rely on deep learning models trained on massive datasets to "infer" or "estimate" BMD values based on image textures and grayscale. This process is essentially a "black box," where the source of the data cannot be traced, and the method lacks a solid physical foundation.
ii.Bone's QCT (Measuring Data): While we do not use external phantoms, our system uses rigorously validated self-referencing phantom technology. This is still grounded in true physical density measurement, ensuring that every data point is traceable. It achieves accuracy equivalent to traditional phantom-based QCT.
(2)Different Clinical Operations: AI-based bone density products do not comply with ISCD standards
Pure AI products perform fully automated calculations for entire vertebral bodies, creating significant clinical issues:
i.Interference from the Basivertebral Vein: The posterior portion of vertebrae contains venous structures (e.g., the basivertebral vein). AI systems calculate the entire vertebra, often including blood, bone islands, and even cortical bone in the same measurement as cancellous bone density, leading to severely distorted data.
ii.Non-adjustable ROI: AI-based systems typically do not allow physicians to manually adjust the ROI (region of interest). In contrast, Bone's QCT follows the standards of the International Society for Clinical Densitometry (ISCD), allowing doctors to manually adjust the ROI to measure only the anterior one-third of the cancellous bone in the vertebra, thus avoiding interference from the basivertebral vein and ensuring pure cancellous bone density measurements.
(3)Different Compliance: AI-based bone density products do not meet clinical diagnostic standards
According to the 2018 Guidelines for the Diagnosis of Osteoporosis with Quantitative CT, the final recommendation specifies that products meant for osteoporosis diagnosis must demonstrate consistency with Mindways QCT. Pure AI products typically fail to meet this requirement.

Bone's QCT has not only undergone comparison validation with Mindways but has also completed multicenter clinical trials. In contrast to competing products whose scientific papers are typically based on single-center trials with selectively curated data that may introduce bias, Bone's QCT has undergone rigorous research and validation. It is fully compliant with relevant diagnostic guidelines and can be confidently used for clinical diagnosis.
Most AI-based products have not conducted validation studies against Mindways. Their registration certificates usually only state that they "can be used for bone density measurement," which is entirely different from "being suitable for diagnostic purposes." Products that fail to meet guideline requirements cannot be used as a basis for clinical diagnosis.
-
Clinical Application Data Transmission
-
What are the data connection methods for Bone's QCT?
(1)Direct connection to the CT machine
(2)Connection via PACS
(3)Using a shared folder
-
Can Bone's QCT connect to PACS?
(1)Bone's QCT can connect to a hospital's PACS system, enabling a single QCT product to correspond to multiple CT devices, support cross-campus data sharing, and remain compatible with various CT and PACS brands.
(2)Differences in hospital PACS systems often lead to poor compatibility for imported brands and smaller manufacturers' products. Even if technically feasible, their actual on-site performance may be disappointing. Bone's technical team has completely overcome this challenge, achieving seamless clinical implementation of high-quality technology. With its strong technical capabilities, Bone's can quickly respond to and address issues, providing hospitals with a stable and efficient after-sales service experience.
-
How is Bone's QCT installed in hospitals? How is CT data retrieved?
For in-hospital deployment, we offer two solutions:
(1)Single-user version: Typically used for small-scale usage, it can be deployed on an in-hospital computer or a Bone's QCT workstation, allowing one user to access the system at the same time.
(2)Multi-user version: Suitable for large-scale usage, it requires a customized server installed within the hospital. Our engineers provide on-site support to connect and deploy the system across multiple hospital computers, enabling simultaneous access for multiple users.
For accessing in-hospital CT data, we offer two solutions:
(1)PACS connection: As the most common approach, upon obtaining hospital authorization, our engineers assist in connecting to the PACS database. Examination data can be directly downloaded, and reports can be directly transmitted back to PACS.
(2)Direct CT machine connection: For situations where PACS connection is not feasible, we support direct CT machine connection, allowing examination reports to either be uploaded to PACS or stored in a local database for convenient review and analysis.
If neither of the above methods is applicable, local data loading can be used for analysis with reports saved locally for future reference.
-
Pricing and Medical Insurance
-
Can QCT bone density measurement be billed?
QCT bone density measurement can be billed in compliance with regulations. The National Medical Products Administration (NMPA) explicitly classifies QCT as computer-aided diagnostic/analysis software, making it an approved category for clinical billing. Currently, billing entries exist nationwide under the name "Bone Mineral Density Measurement." In some provinces, the service is billed through CT under the entry "X-ray Computed Tomography (CT) Scanning." The average nationwide fee for a single site is approximately 100 yuan.
The core of billing lies in providing effective medical diagnostic services. According to the Guidelines for the Diagnosis of Osteoporosis with Quantitative CT (2018 Edition), only devices that have undergone rigorous clinical validation and demonstrated high consistency with Mindways can deliver results used as the basis for clinical diagnosis. Devices that have not undergone clinical trials or consistency validation can only provide data for reference, not for issuing diagnostic conclusions, and therefore do not qualify for medical billing.
Bone's QCT has been validated through large-scale multicenter clinical trials and fully meets guideline requirements. Its results can be directly used for clinical diagnosis, making the billing both compliant and fully justified.
-
Bone’s Manufacturer
-
Why is Bone's capable of developing QCT?
Bone's research and development began in 1994 and has progressed through a complete in-house development cycle, including theory formulation, validation, and industrialization.
The founder of Bone's, Professor WeiJia Lu, is the founder of the Orthopedic Laboratory at the University of Hong Kong. Under the leadership of Academician Professor Chi-yan Leong (John), over 400 scientific papers have been published, with total R&D investment exceeding 100 million yuan.
Bone's R&D team consists of top scholars from around the world, led by Professor WeiJia Lu of the University of Hong Kong and Professor Zhaoyang Li of Tianjin University. The core team members are PhDs and postdoctoral researchers from renowned universities in China and abroad, showcasing strong research capabilities.
-
What is the brand value of Bone's?
Ensuring every elderly individual has healthy and happy bones
-
Development History of Bone's
In 2018, Professor Wei-Jia Lu and Professor Chaoyang Li co-founded Bone's, securing HK$5 million in investment from HKX. In 2020, Bone's established operations in mainland China to pursue medical device registration and built a comprehensive quality and R&D system. By 2022, the company received tens of millions of USD in investment from Sequoia China. In 2023, Bone's secured three medical device registrations and launched the world's first full-body bone density self-referencing phantom calibration technology. By 2024, Bone's achieved nationwide sales in the tens of millions and established scientific collaborations with over 10 renowned tertiary hospitals in China, publishing more than a dozen SCI papers in the field.
-
Future Product Lines of Bone's
Bone's future lies in expanding its medical imaging bone analysis software functionalities further. The company plans to venture into minimally invasive intelligent surgery and the field of drug-device combination consumables. These lines of development include personalized implants, drug-device consumables, AI-assisted orthopedic surgery planning, and customized surgical tools.
-
Other FAQ
-
Why wasn’t QCT popular before?
Previously, QCT in China lacked a Chinese-specific database and clinical guidelines. Over the past few years, these systems have gradually been developed, creating new opportunities for promotion. In general, medical devices in China require nearly 10 years of education and system establishment before reaching widespread acceptance, after which it becomes an ideal time for the adoption of domestic alternatives.
-
Is all QCT technology the same?
Not all QCT systems are the same. Bone’s QCT uses phantommeless synchronized detection, which allows for the examination of historical data. Bone’s bone density system supports multi-machine CT usage simultaneously ("one-to-N"), enabling the integration of all hospital CT scanners, while ensuring patient data security and compliance with hospital information safety protocols.
Additionally, while QCT relies on algorithms, the quality of these algorithms differs. Bone’s QCT provides validated results consistent with Mindways, a recognized standard. Importantly, reliable QCT diagnostic results require two essential elements: a Chinese-specific database and quality control processes, both of which Bone’s uniquely delivers as a domestic brand.
How does Bone’s QCT compare to other products? Bone’s QCT stands out by offering four unique advantages: a Chinese-specific database, local technical support, a comprehensive quality control system, and the ability to produce diagnostic reports. These features make Bone’s QCT the only domestic brand capable of meeting all these key requirements.